 /PeriodicTableCharge-BW-56a12db13df78cf772682c34.png)
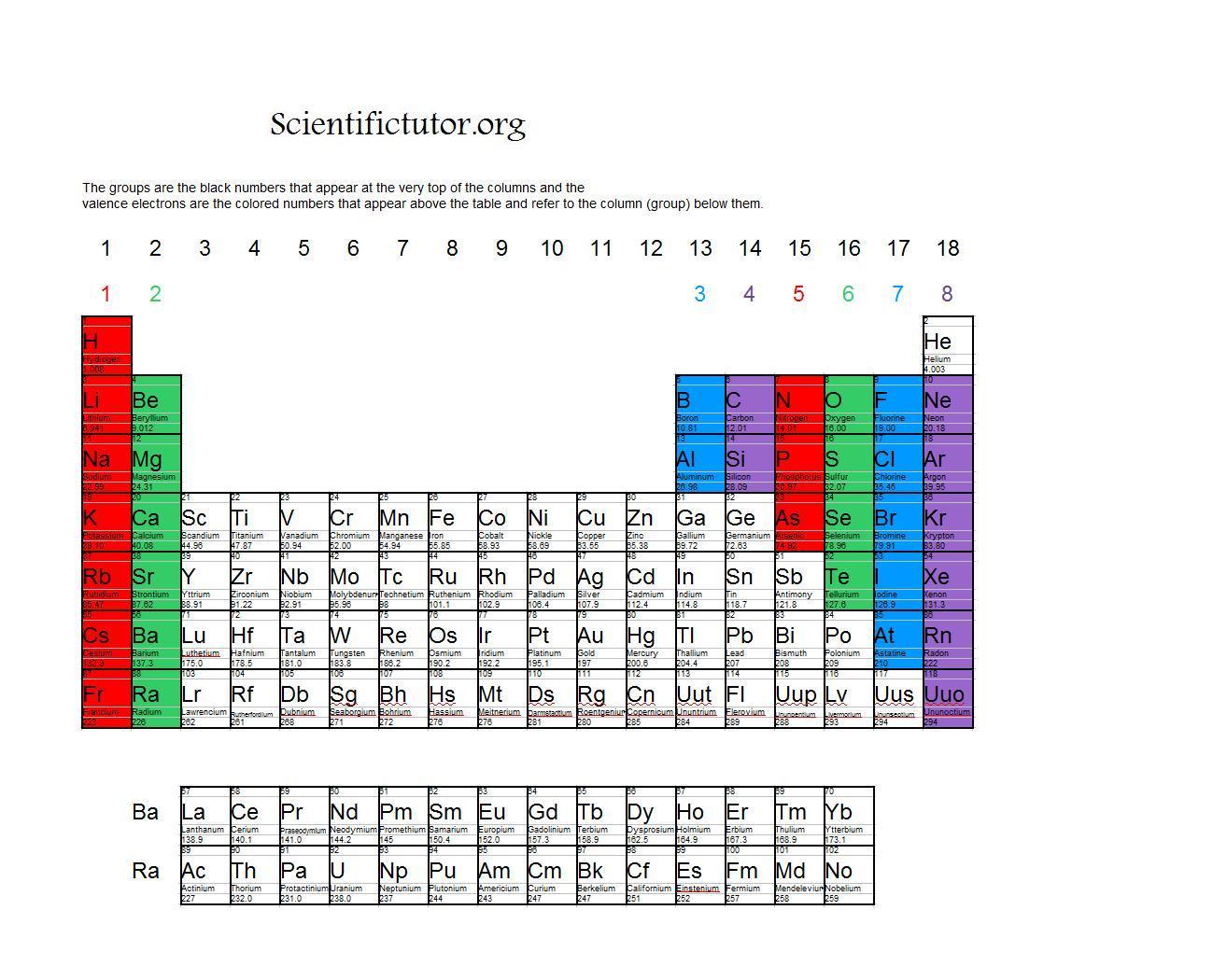
A nonmetal is typically dull and a poor conductor of electricity and heat. Metals are also malleable (they can be beaten into thin sheets) and ductile (they can be drawn into thin wires). A metal is a substance that is shiny, typically (but not always) silvery in color, and an excellent conductor of electricity and heat. We hope you enjoyed studying this lesson and learned something cool about Valence Electrons in Beryllium! Join our Discord community to get any questions you may have answered and to engage with other students just like you! Don’t forget to download our App to experience our fun VR classrooms – we promise it makes studying much more fun□ REFERENCE No, Beryllium does not contain 6 valence electrons. We can use this method to predict the charges of ions in ionic compounds. For example, fluorine has seven valence electrons, so it is most likely to gain one electron to form an ion with a 1- charge. How Many Valence Electrons Does Beryllium Lose?īeryllium could lose 2 valence electrons. When forming ions, elements typically gain or lose the minimum number of electrons necessary to achieve a full octet. From the elements position on the periodic table, predict the valence shell electron configuration for each atom. Tins electron configuration is Kr5 s 2 4 d 10 5 p 2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
